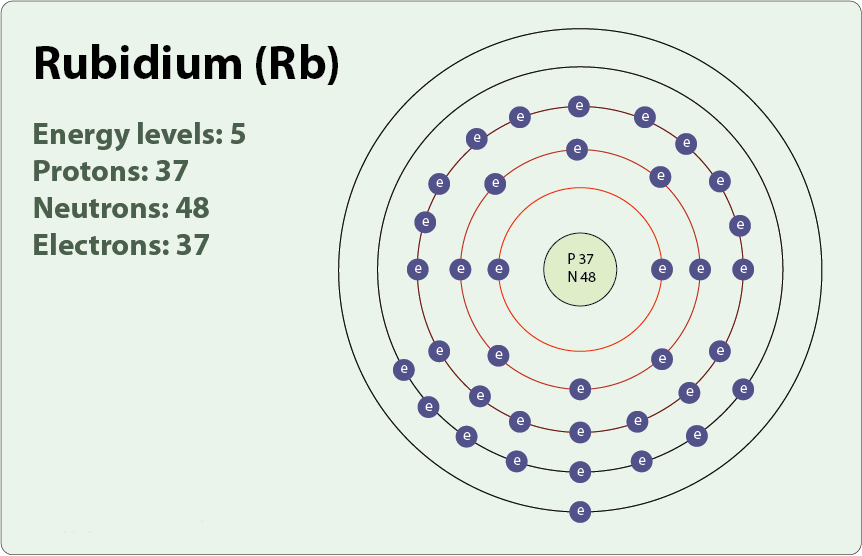
 There have been experiments in which blisters burst where it is stored (bottom image) to fall and explode in bathtubs. It also reacts with it more explosively compared to lithium, sodium and potassium. It is the first of the alkali metals that, despite being not very dense, sinks in water. It is one of the most reactive metals that exist. Its name sounds similar to ruby, and it is because when it was discovered its emission spectrum showed characteristic lines of deep red color. The rubidium It is a metallic element that belongs to group 1 of the periodic table: that of the alkali metals, being represented by the chemical symbol Rb. Decay scheme of K-Ar, U-Pb and Sm-Nd, petrogenetic implications-part A Content 
Potassium is the most reactive metal on the periodic table.Īs of now, noble gases are the least reactive metals.Īs the name itself states, reactivity series is defined as the decreasing order of reactivity in metals of the periodic table.Video: M-09. It is budget-friendly with many benefits, check it out today. Besides, if you have any trouble understanding certain concepts of Chemistry, or any other subjects, then it would be a good idea for you to join online interactive classes offered by Tutoroot. Hope this article above helped you understand all about the metal reactivity series, most reactive elements, features of reactivity series, and applications of reactivity series.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
